Summary
TAVNEOS, in combination with a rituximab or cyclophosphamide regimen, is indicated for the treatment of adult patients with severe, active granulomatosis with polyangiitis (GPA) or microscopic polyangiitis (MPA)2
TAVNEOS should be initiated and monitored by healthcare professionals experienced in the diagnosis and treatment of GPA or MPA2
TAVNEOS-based regimen


achieved non-inferior clinical remission at Week 26 and superior sustained clinical remission at Week 52 vs a GC-based regimen1*

includes TAVNEOS taken as a fixed oral dose2

The most common adverse reactions are nausea (23.5%), headache (20.5%), white blood cell count decreased (18.7%), upper respiratory tract infection (14.5%), diarrhoea (15.1%), vomiting (15.1%), and nasopharyngitis (15.1%). 2

TAVNEOS is the first targeted treatment for GPA/MPA recognised by EULAR and KDIGO3,4,6,7
References & footnotes
Footnotes
*Clinical remission in the ADVOCATE study was defined as a BVAS 0 and no GC use in previous 4 weeks.1
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; BVAS, Birmingham Vasculitis Activity Score; EULAR, European Alliance of Associations for Rheumatology; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; KDIGO, Kidney Disease: Improving Global Outcomes; MOA, mechanism of action; MPA, microscopic polyangiitis.
References
- Jayne D, et al. N Engl J Med 2021;384(7):599–609.
- TAVNEOS EU SmPC January 2025.
- Bekker P, et al. PLoS One 2016;11(10):e0164646.
- European Medicine Agency (2021). First-in-class medicine recommended for treatment of rare blood vessel inflammation. Available at: https://www.ema.europa.eu/en/news/first-class-medicine-recommended-treatment-rare-blood-vessel-inflammation. Date accessed: January 2025.
- Stone J, et al. Semin Arthritis Rheum 2022;55:152010.
- Hellmich B, et al. Ann Rheum Dis 2023;0:1–18.
- Kidney Disease: Improving Global Outcomes (KDIGO) ANCA Vasculitis Work Group. Kidney Int 2024;105(3S):S71–S116.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
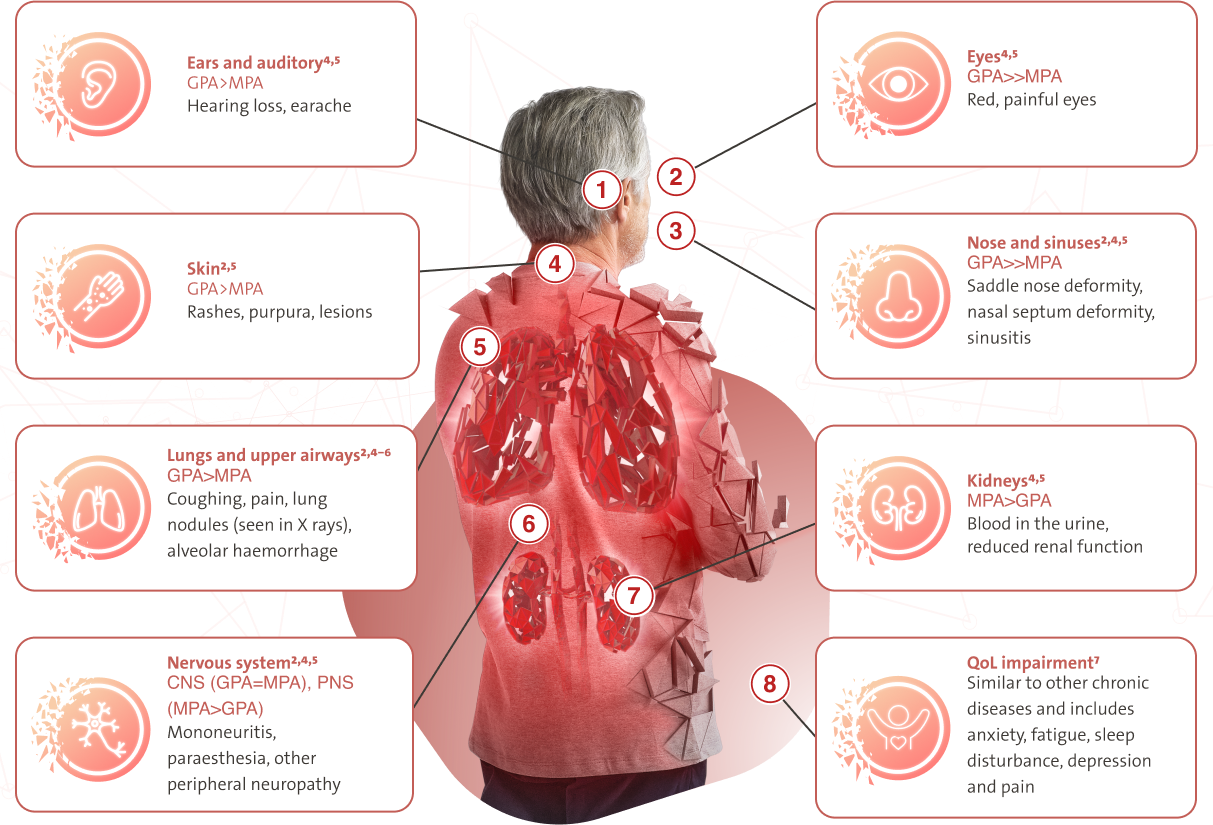
About AAV
HOW DO YOUR AAV PATIENTS PRESENT?

AAV (GPA/MPA) is a severe, relapsing disease that can cause irreversible damage to multiple organs2–4
THERE IS A NEED FOR A NEW AND MORE TARGETED APPROACH TO AAV (GPA/MPA) TREATMENT9–12


AAV (GPA/MPA) is a severe, relapsing disease that can cause irreversible damage to multiple organs2–4

Despite treatment, many AAV patients do not achieve or sustain remission and the risk of relapse persists, further exacerbating organ damage13–15

Established therapies for AAV (GPA/MPA) have relied on non-specific immunosuppressants alongside high-dose and/or long-term GCs, which add to the burden of disease12,15–19

Activation of the alternative complement pathway plays a key role in AAV, and no established therapies address this as a key driver of vascular inflammation10,12,20
References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; CNS, central nervous system; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; MOA, mechanism of action; MPA, microscopic polyangiitis; PNS, peripheral nervous system; QoL, quality of life.
References
- TAVNEOS EU SmPC January 2025.
- Wallace ZS, Miloslavsky EM. BMJ 2020;368:m421.
- Pagnoux C. Eur J Rheumatol 2016;3(3):122–33.
- Al Hussain T, et al. Adv Anat Pathol 2017;24(4):226–34.
- Hunter RW, et al. BMJ 2020;369:m1070.
- Kitching AR, et al. Nat Rev Dis Primers 2020;6(1):71.
- Quartuccio L, et al. J Autoimmun 2020;108:102397.
- Basu N, et al. Ann Rheum Dis 2014;73(1):207–11.
- Hutton HL, et al. Semin Nephrol 2017;37(5):418–35.
- Jennette JC, Nachman PH. Clin J Am Soc Nephrol 2017;12(10):1680–91.
- Bekker P, et al. PLoS One 2016;11(10):e0164646.
- Lamprecht P, et al. EMJ Rheumatol 2021;8(1):36–42.
- Stone JH, et al. N Engl J Med 2010;363(3):221–32.
- Specks U, et al. N Engl J Med 2013;369(5):417–27.
- Robson J, et al. Rheumatology (Oxford) 2015;54(3):471–81.
- Yates M, et al. Ann Rheum Dis 2016;75(9):1583–94.
- Little MA, et al. Ann Rheum Dis 2010;69(6):1036–43.
- McGregor JG, et al. Clin J Am Nephrol 2012;7(2):240–7.
- Turnbull J, Harper L. Best Pract Res Clin Rheumatol 2009;23(3):391–401.
- Liberman AC, et al. Front Endocrinol (Lausanne) 2018;9:235.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Efficacy
Primary Endpoint
IMPROVED DISEASE CONTROL

TAVNEOS-based regimen achieved non-inferior clinical remission at Week 26 and superior sustained clinical remission at Week 52 vs a GC-based regimen2*
At Week 26, a TAVNEOS-based regimen demonstrated non-inferiority in achieving clinical remission vs a GC-based regimen2
p<0.001 for non-inferiority (Difference 3.4, 95% CI, -6.0 to 12.8)

At Week 52, a TAVNEOS-based regimen demonstrated superior sustained clinical remission vs a GC-based regimen2
p=0.007 for superiority (Difference 12.5, 95% CI, 2.6 to 22.3)


At Week 52, TAVNEOS-based regimen demonstrated a number-needed-to-treat (NNT) between 8 and 10†
ADVOCATE STUDY DESIGN

52 week, international, multicentre, active-comparator, randomised, double-blind, double-dummy, controlled, pivotal phase 3 study with patients assigned to receive TAVNEOS 30 mg twice daily (N=166) or prednisone tapering schedule for 20 weeks (60 mg per day tapered to discontinuation by Week 21) (N=164) on the top of either cyclophosphamide (followed by azathioprine) or rituximab.2
TAVNEOS-based regimen patients received NO study-supplied GCs. Patients in both regimens could use non-study supplied GCs throughout the 52-week study at their physician’s discretion for AAV worsening or non-AAV-related reasons. Non-study supplied GCs during the screening had to be tapered to ≤20 mg per day before entering the study, and further tapered to 0 mg by the end of Week 4 of the study.5
Key inclusion and exclusion criteria5
Inclusion criteria
- ≥12 years old*
- Newly-diagnosed or relapsing GPA or MPA according to Chapel Hill Consensus Conference definitions
- Indicated for RTX or CYC treatment
- Either MPO+ or PR3+
- Estimated eGFR of ≥15 mL/min/1.73 m2
- At least one major or 3 non-major items, or at least two renal items of haematuria and proteinuria on BVAS
Exclusion criteria
- Alveolar haemorrhage requiring invasive pulmonary ventilation anticipated to last beyond screening
- Any other multisystem autoimmune disease
- Coagulopathy or bleeding disorder
- Required dialysis or plasma exchange within 12 weeks prior to screening
- A kidney transplant
- Had received CYC within 12 weeks prior to screening, RTX within 12 months prior to screening (or 6 months with B cell reconstitution, CD19 count >0.01x109/L), cumulative dose of IV GCs >3 g within 4 weeks, or oral GCs of >10 mg per day prednisone (or equivalent) for >6 weeks continuously prior to screening
*Adult and adolescent GPA/MPA patients participated in this study. For additional details, visit clinicaltrials.gov, study code: NCT02994927
Baseline characteristics2
Primary endpoints2
References & footnotes
Footnotes
*Clinical remission in the ADVOCATE study was defined as a BVAS 0 and no GC use in previous 4 weeks.2
†The NNT was calculated using https://www.statology.org/confidence-interval-difference-in-proportions-calculator/ to determine the confidence intervals for crude difference in proportions. At Week 52, the response rates were 65.7% in the TAVNEOS-based regimen and 54.9% in the GC-based regimen. This led to a crude result of NNT = 1/(0.657-0.549) = 9.26, rounded to 10 patients. Using the adjusted model and the estimated common difference, the NNT = 1/0.125 = 8. In conclusion, the NNT is between 8 and 10 patients at Week 52.2
Abbreviations
AAV, ANCA-associated vasculitis; AIS, Aggregate Improvement Score; ANCA, anti-neutrophil cytoplasmic antibody; BVAS, Birmingham Vasculitis Activity Score; CD19, cluster of differentiation 19; CI, confidence interval; CKD, chronic kidney disease; CWS, Cumulative Worsening Score; CYC, cyclophosphamide; eGFR, estimated glomerular filtration rate; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; GTI, Glococorticoid Toxicity Index; IV, intravenous; MCP-1, monocyte chemoattractant protein-1; MOA, mechanism of action; MPA, microscopic polyangiitis; MPO, myeloperoxidase; NNT, number-needed-to-treat; PR3, proteinase-3; RTX, rituximab; SD, standard deviation; SF-36, Medical Outcomes Survey Short-Form 36; UACR, urine albumin-to-creatinine ratio; VAS, Visual Analogue Scale.
References
- TAVNEOS EU SmPC January 2025.
- Jayne D, et al. N Eng J Med 2021; 384(7): 599–609.
- Bekker P, et al. PLoS One 2016;11(10):e0164646.
- European Medicine Agency (2021). First-in-class medicine recommended for treatment of rare blood vessel inflammation. Available at: https://www.ema.europa.eu/en/news/first-class-medicine-recommended-treatment-rare-blood-vessel-inflammation. Date accessed: January 2025.
- Jayne D, et al. N Engl J Med 2021;384(7):599–609. [Supp Appendix].
- TAVNEOS EPAR. Available at: https://www.ema.europa.eu/en/documents/assessment-report/tavneos-epar-public-assessment-report_en.pdf. Date accessed: January 2025.
- Cortazar F, et al. Kidney Int Rep 2023;8(4):860–70.
- Jayne D, et al. J Am Soc Nephrol 2021:32.
- NHS (2019). Chronic Kidney Disease. Available at: https://www.nhs.uk/conditions/kidney-disease/diagnosis/. Date accessed: January 2025.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Safety & dosing
SAFETY PROFILE

The most common adverse reactions observed in a pivotal Phase 3 study in patients treated with a TAVNEOS-based regimen:1

Nausea
23.5%

Headache
20.5%

Decreased white
blood cell count
18.7%

Nasopharyngitis
15.1%

Diarrhoea
15.1%

Vomiting
15.1%

Upper respiratory
tract infection
14.5%

The most common serious adverse reactions are liver function abnormalities (5.4%) and pneumonia (4.8%)1
In post marketing setting: drug-induced liver injury and vanishing bile duct syndrome have been reported (frequency unknown)1

TAVNEOS is a substrate of CYP3A4, but clinically relevant interactions are unlikely when TAVNEOS is co-administered with inducers or inhibitors of this enzyme1
COMPARABLE SAFETY RESULTS

Numerically similar incidence of SAEs observed in TAVNEOS- and GC-based regimen2

In a pooled safety analysis, the AE patient first incidence rate, AE rate, SAE rate, infection event rate, and WBC count decrease AE rate were statistically lower in the TAVNEOS-based regimen vs GC-based regimen3
FIXED ORAL DOSING & MONITORING

TAVNEOS is taken as a fixed oral dose, with required monitoring1
TAVNEOS should be taken as follows:1

30 mg TAVNEOS

Taken twice-daily

With food
Patients must be monitored for:
- Hepatic transaminases and total bilirubin at least every 4 weeks after the start of therapy for the first 6 months of treatment, and as clinically indicated thereafter1*
- White blood cell count as clinically indicated and as part of the routine follow-up of the patient’s underlying condition1†

Treatment regimen in combination with immunosuppressants (RTX or CYC/AZA)1
References & footnotes
Footnotes
*Treatment must be re-assessed clinically and temporarily stopped if ALT or AST is >3x ULN. Treatment must be temporarily stopped if ALT or AST is >5× ULN. Please consult Summary of Product Characteristics for information about permanent discontinuation.1
†Treatment must be temporarily stopped if a patient develops leukopenia (white blood cell count <2×109/L) or neutropenia (neutrophils <1×109/L) or Lymphopenia (lymphocytes <0.2×109/L).1
Abbreviations
AAV, ANCA-associated vasculitis; AE, adverse event; ALT, alanine transaminase; ANCA, anti-neutrophil cytoplasmic antibody; AST, aspartate transaminase; AZA, azathioprine; CYC, cyclophosphamide; CYP3A4, Cytochrome P450 3A4; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; MOA, mechanism of action; MPA, microscopic polyangiitis; RTX, rituximab; SAE, serious adverse event; TEAE, treatment-emergent adverse event; ULN, upper limit of normal; WBC, white blood cell.
References
- TAVNEOS EU SmPC January 2025.
- Jayne D, et al. N Engl J Med 2021;384(7):599–609.
- Jayne D, et al. J Am Soc Nephrol 2022:33.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
MOA
FIRST-IN-CLASS TARGETED TREATMENT

TAVNEOS is the only targeted therapy for AAV (GPA/MPA) to address a key driver of vascular inflammation in the alternative complement pathway2-4

Activation of the alternative complement pathway plays a key role in AAV, and no established therapies address this as a key driver of vascular inflammation2,5-10
TREATMENT RECOGNISED BY EULAR 2022 AND KDIGO 2024

Both KDIGO 2024 and EULAR 2022 guidelines recognize avacopan (Tavneos) as an important glucocorticoid‑sparing option for remission induction in ANCA‑associated vasculitis (AAV). Both recommend that avacopan may be used in combination with rituximab (RTX) or cyclophosphamide (CYC), especially in patients at high risk of glucocorticoid toxicity.

Eular 2022 recommendation level of evidence 1b:
TAVNEOS (avacopan) in combination with RTX or CYC may be considered for induction of remission in GPA or MPA, as part of a strategy to substantially reduce exposure to GCs11

KDIGO 2024 PRACTICE POINTS 9.3.1.1 AND 9.3.1.7:
TAVNEOS may be used as an alternative to GCs for induction of remission in combination with RTX or CYC12*
Understand more about the pathogenesis of AAV and how TAVNEOS works
References & footnotes
Footnotes
*Recommendation 9.3.1.1: We recommend that GCs in combination with RTX or CYC be used as initial treatment of new-onset AAV (1B). Practice Point 9.3.1.7: Avacopan may be used as an alternative to GCs. Patients with an increased risk of GC toxicity are likely to receive the most benefit from avacopan.
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; C5a, complement component 5a; C5b, complement component 5b; C5L2, C5a receptor-like 2; CYC, cyclophosphamide; EULAR, European Alliance of Associations for Rheumatology; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; KDIGO, Kidney Disease: Improving Global Outcomes; MAC, membrane attack complex; MOA, mechanism of action; MPA, microscopic polyangiitis; RTX, rituximab.
References
- TAVNEOS EU SmPC January 2025.
- Bekker P, et al. PLoS ONE 2016;11(10):e0164646.
- Moiseev S, et al. Clin Exp Immunol 2020;202(3):394–402.
- European Medicine Agency (2021). First-in-class medicine recommended for treatment of rare blood vessel inflammation. Available at: https://www.ema.europa.eu/en/news/first-class-medicine-recommended-treatment-rare-blood-vessel-inflammation. Date accessed: January 2025.
- Hutton HL, et al. Semin Nephrol 2017;37(5):418–35.
- Jennette JC, Nachman PH. Clin J Am Soc Nephrol 2017;12(10):1680–91.
- Al-Hussain T, et al. Adv Anat Pathol 2017;24(4):226–34.
- Lamprecht P, et al. EMJ Rheumatol 2021;8(1):36–42.
- Liberman AC, et al. Front Endocrinol (Lausanne) 2018;9:235.
- Nozaki Y. Front Immunol 2021;12:631055.
- Hellmich B, et al. Ann Rheum Dis 2023;0:1–18.
- Kidney Disease: Improving Global Outcomes (KDIGO) ANCA Vasculitis Work Group. Kidney Int 2024;105(3S):S71–116.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Resources
References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; GPA, granulomatosis with polyangiitis; MOA, mechanism of action; MPA, microscopic polyangiitis
References
- TAVNEOS EU SmPC January 2025.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
DK-NA-2600005 | Date of preparation: April 2026
Report an Adverse Event. CSL collects information on adverse events to be able to monitor product safety and to investigate, evaluate and report adverse effects to applicable authorities.
To report an adverse event, contact us here or report via the Danish Medicines Agency (Lægemiddelstyrelsen).
When you report a suspected adverse event, we collect certain personal data. If you submit a report within the EU, you can read more about how we handle your personal data here: Privacy Notice for Reporting Suspected Side Effects within Europe and GDPR.